Page added on January 10, 2014
Tepco “Explains” Plumes Of Smoke Above Exploded Fukushima Reactor 3
In the past two weeks there was significant speculation that the Fukushima catastrophe was once again rapidly escalating after plumes of mysterious smoke were detected above the destroyed Reactor 3 – the one with a mixed MOX and uranium fuel core. RT described it as follows:
Fresh plumes of most probably radioactive steam have been detected rising from the reactor 3 building at the crippled Fukushima nuclear plant, said the facility’s operator company.
The steam has been detected by surveillance cameras and appeared to be coming from the fifth floor of the mostly-destroyed building housing crippled reactor 3, according to Tokyo Electric Power Co (TEPCO), the plant’s operator.
The steam was first spotted on December 19 for a short period of time, then again on December 24, 25, 27, according to a report TEPCO published on its website.
The company, responsible for the cleanup of the worst nuclear disaster since Chernobyl, has not explained the source of the steam or the reason it is rising from the reactor building. High levels of radiation have complicated entry into the building and further inspection of the situation.
Naturally, fears arose that more uncontrolled meltdowns were in process at the exploded reactor.
This morning, probably in order to assuage fears that it has lost control more than usual, TEPCO released the following statement “Regarding Certain Overseas Reports on Fukushima Daiichi Nuclear Power Station.” TEPCO’s soothing words – “We found no abnormalities in the measured values (indicating the temperature and condition of the Reactor Building), or in the value of the monitoring post (monitoring the amount of radiation), even when the steam-like gas was being generated, therefore we are certain that there has been no influence on the outside. In addition, we measured the amount of radiation at the point from which steam-like gas was generated, and found that its amount was almost the same as for the other neighboring points.”
And as everyone knows by now, TEPCO would never lie.
Full statement from the semi-nationalized company:
Regarding Certain Overseas Reports on Fukushima Daiichi Nuclear Power Station
Some overseas press outlets are reporting that steam is being generated from Unit 3 at Fukushima Daiichi NPS and its condition is dangerous, releasing radioactive material, and that there were two underground nuclear explosions at the site. However, such information is incorrect, and we have found no change in the status of the plant.
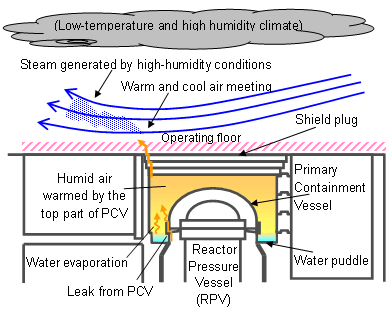
Steam generation on the operating floor at Unit 3, and the detection of highly concentrated radioactive material at groundwater observation holes etc., have been pointed out as the basis for the overseas reports. We respond to these as follows.
– Steam-like gas generated on the operating floor at Unit 3 in Fukushima Daiichi NPS
Since July 2013, steam-like gas has been intermittently observed on the operating floor at Unit 3. The steam-like gas is estimated to emerge via the following sequence.
1) Accumulated hygroscopic moisture, such as rainwater, exists below the shield plug (a lid made of concrete).
2) The hygroscopic moisture is heated by the heat radiated from the top of the Primary Containment Vessel (PCV).
3) The heated hygroscopic moisture is released onto the operating floor via the gap of the shield plug.
4) The released moisture is cooled down by the cool atmosphere, giving it the appearance of a steam-like gas.
We found no abnormalities in the measured values (indicating the temperature and condition of the Reactor Building), or in the value of the monitoring post (monitoring the amount of radiation), even when the steam-like gas was being generated, therefore we are certain that there has been no influence on the outside.
In addition, we measured the amount of radiation at the point from which steam-like gas was generated, and found that its amount was almost the same as for the other neighboring points.

– Highly-concentrated radioactive material found at the observation holes
We have been monitoring the groundwater sampled at the observation holes established to investigate the contamination status of groundwater, for the purpose of investigating the effects of the leak of contaminated water from the contaminated water storage tanks in August 2013. At the end of 2013, the measurement value for Tritium, one of the radioactive materials in the water sampled at the observation hole near the tank that suffered leakage, increased from 34,000Bq/L (on December 28) to 450,000Bq/L (on January 1). This increase in the value could be attributed to 1) the contaminated water that previously leaked from the tank soaking into the nearby ground, or 2) the effects of the water pumping* (*we have been pumping up the contaminated groundwater at the nearby observation holes, however the amount being pumped up was temporarily decreased at the end of 2013). The value decreased to 17,000Bq/L on January 8. Highly concentrated tritium (almost equivalent concentration level) was found in this observation hole in the past, and the highest concentrated tritium 790,000Bq/L was also found here on October 17, 2013.
– Earthquake on December 31, 2013
Some overseas press outlets reported that underground nuclear explosions caused several quakes with magnitudes of 5.1 and 3.6.
According to the Japanese Meteorological Agency, 13 earthquakes (with a maximum magnitude of 5.4) occurred and originated in the north part of Ibaraki Prefecture on December 31 2013. None of these earthquakes was caused by Fukushima Daiichi NPS. We also found no accidents or trouble etc. in Fukushima Daiichi NPS.
30 Comments on "Tepco “Explains” Plumes Of Smoke Above Exploded Fukushima Reactor 3"


J-Gav on Fri, 10th Jan 2014 3:59 pm
TEPCO are a lying sack of green S-h^@qit.
foxv on Fri, 10th Jan 2014 4:39 pm
Interesting thing about lying all the time, is eventually the truth comes out. Time will tell all at some point.
Interesting that the article says:
” moisture is heated by the heat radiated from the top of the Primary Containment Vessel (PCV).”
yet the diagram has the label “Leak from PCV”
oops, that wouldn’t happen to be a bit of truth accidentally slipping out would it?
Kenz300 on Fri, 10th Jan 2014 5:22 pm
TEPCO was not honest and open about this disaster from day one.
They can not be trusted to be honest and open now.
There needs to be more independent monitoring and reporting of this disaster.
Davy, Hermann, Mo on Fri, 10th Jan 2014 6:00 pm
Is TEPCO telling white lies or serious lies? Sounds pathological and kind of like desperate dictator ministers sound after a while. Remember the Iraqi information minister claiming the Americans were 200 miles from Bagdad when they were actually at the outskirts or Hitler’s Germany near the end!
I used to think the financial system coupled with energy issues would bring on a collapse of some degree now I am wondering if the Fukushima rolling snow ball will be the gasoline on the fire.
chilyb on Fri, 10th Jan 2014 6:48 pm
this is no worse than spilling a few thousand tons of bananas.
GregT on Fri, 10th Jan 2014 7:17 pm
“this is no worse than spilling a few thousand tons of bananas.”
I guess that’s why all of those stupid people are willing to spend tens of billions of dollars over the next 40 years just to get the site contained, and most likely tens of billions of dollars more over the next few centuries in an attempt to keep it that way.
Yup, just like bananas.
Kenz300 on Fri, 10th Jan 2014 7:24 pm
Nuclear energy —they said is was too cheap to meter…….. now we find out it is too costly and too dangerous for humanity.
We can not trust the entrenched nuclear interests to tell us the truth. They will do all they can to protect their profits and their industry. Outside international inspectors that are truly independent need to be brought in to monitor the site and provide truthful information to the world.
GregT on Fri, 10th Jan 2014 7:28 pm
kenz,
I don’t think that the world is ready for the truth……………..
Hugh Culliton on Fri, 10th Jan 2014 11:25 pm
“this is no worse than spilling a few thousand tons of bananas.”
If those bananas happen to be made of highly radioactive MOX capable of sterilizing the entire Pacific Ocean. But you were just joking, right? Because if you weren’t you deserve several trollish comments.
Hugh Culliton on Fri, 10th Jan 2014 11:26 pm
He was joking, Greg.
GregT on Fri, 10th Jan 2014 11:46 pm
Sorry chilyb 🙂 missed that.
Davy, Hermann, Mo on Fri, 10th Jan 2014 11:55 pm
Greg T, “tens of billions of dollars more over the next few centuries in an attempt to keep it that way” that statement may be a long shot….can we manage these toxins for more than 2 decades considering the possibility of a significant collapse in 10 years or less.
I feel much more in the know on energy and somewhat on the financial crisis issues. This Nuk stuff is more difficult to get a handle on.
I am reading and watching videos on it. One of the best sources I have seen down plays some of the worst news going viral right now. I understand the effects of radiation exposure are linear. So even low level exposure adds up. No exposure is good. Old people will have built up far more than younger folks. The speaker is concerned about the buildup of radiation in the food chain but is not as worried about exposure by the beach. So he is avoiding pacific fish.
Well, any help from you all getting a handle on this would be great.
GregT on Sat, 11th Jan 2014 12:34 am
Davy,
“can we manage these toxins for more than 2 decades considering the possibility of a significant collapse in 10 years or less”
I completely agree with you. Even without a ‘significant collapse in 10 years or less’, ( which appears very likely ) there is no doubt that we will run out of fossil fuels far before the nuclear sites will be considered ‘safe’. At the very best, our offspring will be stuck with these messes for a very long time, with less and less resources to deal with them. Hell, we can’t even deal with them now. I find that extremely irresponsible.
There has been plenty of research into the effects of radiation. The problem is, it is very difficult to study something in a controlled environment, when the effects are not seen for several decades. If you haven’t already done so, I would check out some of the more recent reports coming out on the effects from Chernobyl. Some very sobering stuff.
Welch on Sat, 11th Jan 2014 12:59 am
Davy,
May I suggest “Radiation: What It Is, What You Need to Know” by Robert Peter Gale. Very good.
GregT on Sat, 11th Jan 2014 2:33 am
Davy,
Here is a report released by the International Physicians for the Prevention of Nuclear War, ( or the IPPNW) in Germany.
http://www.ippnw.org/pdf/chernobyl-health-effects-2011-english.pdf
An excerpt from the executive summary:
The genetic defects caused by Chernobyl will continue to trouble the world for a long time to
come – most of the effects will not become apparent until the second or third generation.
Even if the extent of the health effects is not yet clear, it can still be predicted that the
suffering brought about by the nuclear disaster in Fukushima is, and will be, of a similar
magnitude.
SilentRunning on Sat, 11th Jan 2014 3:45 am
Hugh Culliton wrote:
> If those bananas happen to be made of highly radioactive MOX capable of sterilizing the entire Pacific Ocean. But you were just joking, right? Because if you weren’t you deserve several trollish comments.
Hugh – I agree with you on several things:
1) Nuclear energy is a fundamentally dangerous technology.
2) That releasing highly radioactive, long lived isotopes into the ocean (or air, or ground) is a really, really bad idea.
3) It is possible to release enough radioactive material into the Pacific ocean to sterilize it – to kill all life that is in the water.
On point #3 – what would you estimate the quantity of radioactive material necessary to in fact sterilize the entire Pacific Ocean? I think you and I can agree that it is more than 1 radioactive atom.
Would 1 pound of spent fuel rods do the job? 10 pounds? 1 ton? 10 tons?
I’m curious as to how you would estimate the quantity of material.
SilentRunning on Sat, 11th Jan 2014 4:04 am
“Sterilizing the Pacific”
From a post on ar15.com:
“It’s estimated that there is about 714 million cubic kilometers of water in the Pacific.
The worst estimate of contaminated water leaking per day from Fukushima is 300 tons. 300 tons of water is 80703 gallons which is 0.0000003055 cubic kilometers.
The daily flow of contaminated water is therefore equal to one part per 1,668,726,667,577,741,000 parts of uncontaminated water.
It is also estimated that the radioactivity of the leaking water is equivalent to 0.6 terabecquerels (trillion becquerels) per month (roughly 50% CS-137 and 50% CS-134), which if distributed equally across the pacific ocean equals 840 becquerels of radiation per cubic kilometer of water, 840 per cubic kilometer equals 0.0000000008403 becquerels per liter of water.
The allowed levels of CS-134 and CS-137 in drinking water is 10 becquerels per liter of water. It would therefore take 1 million years of water leaking from Fukushima for the water in the Pacific to reach levels not considered safe for drinking, and even then you would need to drink 30 liters of contaminated water to even equal a years worth of normal background radiation.
Of course with CS-134 having a half-life of just over 2 years most of the radiation will be gone before it could reach a dangerous level.
I’m not saying the water at the source isn’t dangerously radioactive but the idea that the whole pacific will be contaminated isn’t realistic.”
The above analysis shows that it would take 1 million years for Fukushima leaking radiation to even approach the levels at which it would not be safe to drink – the sterilization level would be significantly higher. But let’s assume that the above facts are from “pro-nuke” sources and that the real level to become unsafe/lethal is really a factor of 100 lower – that would mean it would take 10,000 years of Fukushima leaking to kill the Pacific ocean.
Well, in 10,000 years – all of the radioactive Cesium, Strontium and Tritium will have decayed to non-radioactive forms. So even with generous allowances for safety factors, we can say that Fukushima will not sterilize the entire Pacific ocean.
Will it seriously contaminate areas in and around the reactors? You bet. I wouldn’t eat any sea food that came from 100 miles from the place.
SilentRunning on Sat, 11th Jan 2014 5:09 am
Suppose I am a totally evil mad scientist who wishes to sterilize the entire Pacific ocean for .
Now, I know what I want to do – the question is: How much radioactive Cesium isotopes do I need to obtain to accomplish my sinister goal?
As a previous post noted: The Pacific Ocean is about 714 million cubic kilometers, which is about 7.14e20 liters (714 with 18 zeroes after it) Damn! That’s a lot of liters!
Now, the safe drinking level for Cesium isotope is 10 becquerels per liter. Clearly, I need to go higher than “safe” – I want UNSAFE – lethal levels. I will assume that 30 times higher is needed to kill anything in the water – to *sterilize* it.
Ok, so I want 300 becquerels of radiation per liter, and I need to contaminate 7e20 liters – I need 2.14e23 becquerels of radioactive Cesium.
Now, how many grams of Cesium is that? 1 gram of Cs-137 has an activity level of 3.2e12 becquerels. So I need 6.7e10 grams of Cesium, or 6.7e7 kilograms.
That’s 67 *MILLION* kilograms of Cesium! Over 73 thousand tons!
So how much of spent nuclear fuel rods are radioactive Cesium? Well, if the fuel rod has been in the reactor for the maximum amount of time, it is about 6% cesium. So I need about 1.2 million tons of spent nuclear fuel rods to achieve my goal.
Unfortunately for my wicked scheme – there are less than 2000 tons of spent fuel rods in all of Fukushima. So even if I were to take every scrap of spent fuel at Fukushima, grind it into a fine dust, and spread it all over the Pacific Ocean, I would only get to radiation levels that are a mere 0.2% of my diabolical goal. That wouldn’t even exceed the safe drinking water levels!
Curses! I will have to work on my experimental Death Ray instead!
Makati1 on Sat, 11th Jan 2014 8:21 am
Silent… a plankton absorbs radiation, then a sardine eats 10,000 plankton. That sardine is eaten by a grouper who eats 10,000 sardines. that grouper is eaten by a tuna, that eats 1,000 groupers. That tuna is caught and put on your plate. How much radiation is in that bite of tuna you just swallowed?
chilyb on Sat, 11th Jan 2014 1:44 pm
of course I was joking. I apologize if it wasn’t clear.
Just to clarify my comment – I’m growing weary of hearing justification on how the ongoing radioactive release isn’t going to affect anything in the Pacific due to dilution – and the horseshit I keep reading where comparisons are made between the amount of radioactivity in Cs137 (beta and gamma emitter, 30 year half-life) with that of the naturally occuring Potassium 40 isotope (beta emitter, billion year half-life) is where the bananas comment came from. Technically true, but no bearing on relative mutagenic effects.
What I would like to hear is more about how they plan to remove the thousands of tons of molten fuel that is (currently) sitting about 100 yards from the ocean. Pulling a few dozen of the undamaged rods from reactor 4 isn’t doing it for me. How long do we have to wait before we can enter these structures? And in the meantime, the fuel will be in contact with the groundwater – or perhaps more correctly, an underground river – that is flowing out to the ocean?
SilentRunning on Sat, 11th Jan 2014 4:10 pm
Makati1 wrote:
>Silent… a plankton absorbs radiation, then a sardine eats 10,000 plankton. That sardine is eaten by a grouper who eats 10,000 sardines. that grouper is eaten by a tuna, that eats 1,000 groupers. That tuna is caught and put on your plate. How much radiation is in that bite of tuna you just swallowed?
Two things: 1. Cesium does NOT bio-accumulate. That is, it does not continually build up in organic tissues. Instead, it gets flushed out as part of normal metabolism, like sodium.
2. It is very measurable how much radioactive elements are present in the tuna. If the tuna has ingested a large amount of them – we can know that with instruments. So far, tuna that has been caught a safe distance from Fukushima do not show unsafe levels of cesium-137.
SilentRunning on Sat, 11th Jan 2014 4:26 pm
chilyb wrote:
>I’m growing weary of hearing justification on how the ongoing radioactive release isn’t going to affect anything in the Pacific due to dilution
Radiation *IS* affecting the area around Fukushima – there are unsafe levels of radioisotopes in fish caught in Fukushima harbor. I personally wouldn’t want to eat anything caught within 100 miles of there without extensive testing.
>and the horseshit I keep reading where comparisons are made between the amount of radioactivity in Cs137 (beta and gamma emitter, 30 year half-life) with that of the naturally occuring Potassium 40 isotope (beta emitter, billion year half-life) is where the bananas comment came from. Technically true, but no bearing on relative mutagenic effects.
beta emitters are just as bad as gamma emitters, when you have eaten them.
As for half-life – that really doesn’t matter in this discussion, as it is question of how much radioactive decays per second are occurring.
>What I would like to hear is more about how they plan to remove the thousands of tons of molten fuel that is (currently) sitting about 100 yards from the ocean.
There are not *thousands* of tons of *molten* fuel. There are, perhaps, _tens_ of tons of _melted_ fuel. Words matter in Engineering, physics and in law.
> Pulling a few dozen of the undamaged rods from reactor 4 isn’t doing it for me. How long do we have to wait before we can enter these structures? And in the meantime, the fuel will be in contact with the groundwater – or perhaps more correctly, an underground river – that is flowing out to the ocean?
As the analysis that I posted showed – even if we took all the radioactive debris in Fukushima, dissolved it in powerful acid and then dispersed it in the Pacific ocean, it still would not be enough to “sterilize” the entire ocean.
MOST of the radioactive fuel at Fukushima is still on the site. The used fuel rods in the pools haven’t melted, and haven’t entered the ground water, and haven’t gone into the ocean. They are still – in the fuel pools. A tiny fraction of the fuel in the reactors has melted, has dissolved into the ground water, and has entered the ocean via underground currents. This is very, very bad news for Fukushima harbor and surrounding areas, but it WILL NOT kill everything in the Pacific ocean. Not even close – as the analysis I posted shows. Even liquifying the Fukushima site and spreading it all over the Pacific doesn’t do that.
GregT on Sat, 11th Jan 2014 4:40 pm
“So far, tuna that has been caught a safe distance from Fukushima do not show unsafe levels of cesium-137.”
True, so far the levels detected in Tuna have not been above ‘acceptable’ levels.
http://topinfopost.com/2013/10/10/fukushima-is-here-all-bluefin-tuna-caught-in-california-are-radioactive
There are also concerns in other fish species
http://www.globalresearch.ca/is-fukushima-radiation-contaminating-tuna-salmon-and-herring-on-the-west-coast-of-north-america/5346942
And a recent study has found that “Even the very lowest levels of radiation are harmful to life”
http://www.washingtonsblog.com/2013/08/are-the-levels-of-fukushima-radiation-hitting-north-america-harmless.html
SilentRunning on Sat, 11th Jan 2014 8:41 pm
GregT wrote:
>True, so far the levels detected in Tuna have not been above ‘acceptable’ levels.
http://topinfopost.com/2013/10/10/fukushima-is-here-all-bluefin-tuna-caught-in-california-are-radioactive
Yes, cesium-137 levels in fish have gone up *slightly* since Fukushima. I don’t consider a 2% rise is Cs-137 levels to be particularly alarming. Please get back to me if and when you have some data that shows that levels are spiking to dangerous levels, that would cause fish to be, for example, more radioactive than eating a banana.
>There are also concerns in other fish species
http://www.globalresearch.ca/is-fukushima-radiation-contaminating-tuna-salmon-and-herring-on-the-west-coast-of-north-america/5346942
Yep, there’s some kind of disease affecting salmon right now. Is is caused by Fukushima? Your link raises the question, but does NOTHING to firm up a causal link. Get back to me if you have firm data, for example, showing that affected fish are considerably more radioactive than normal.
>And a recent study has found that “Even the very lowest levels of radiation are harmful to life”
http://www.washingtonsblog.com/2013/08/are-the-levels-of-fukushima-radiation-hitting-north-america-harmless.html
The article makes several dubious claims, and fails to point out that Cesium, just like Potassium, is bio-excreted and doesn’t build up in the body.
chilyb on Sat, 11th Jan 2014 8:44 pm
Silent Running – thanks. I haven’t come across anything that discusses the amount of molten fuel. Would you be able to provide a source? Not disputing your claims, just interested in learning as much as I can.
And any thoughts on a time frame for when they will be able to enter the damaged 1-3 reactor buildings and assess the damage?
SilentRunning on Sun, 12th Jan 2014 12:10 am
chillyb wrote:
>Silent Running – thanks. I haven’t come across anything that discusses the amount of molten fuel. Would you be able to provide a source? Not disputing your claims, just interested in learning as much as I can.
My source is the complete lack of short lived fission products, like Iodine-131, with half life of 8 days. If you detect significant amounts of I-131, then you have fission going on in the last few months. I have heard no reports of I-131.
There is no molten fuel right now, because there is no source of heat energy intense enough to keep fuel in a melted state. Plus, the temperatures reported on the coolant lines are inconsistent with the extremely high temperatures to keep fuel melted.
I grant you that some of the fuel in reactors 1-3 have melted. I don’t know how much, but the upper limit is all of the fuel that was in the reactors at the time of the accident.
>And any thoughts on a time frame for when they will be able to enter the damaged 1-3 reactor buildings and assess the damage?
My educated guesstimate is that there will be robotic inspection in a year or 2.
Kenz300 on Sun, 12th Jan 2014 1:43 am
There is no safe amount of radiation exposure……..
It is cumulative in your life time…….
Keen on Sun, 12th Jan 2014 1:44 am
(1) SilentRunning said: “Cesium, just like Potassium, is bio-excreted and doesn’t build up in the body.”
Wrong.
Here’s the truth:
Cesium-137 can take years to excrete and meanwhile “that’s more than long enough to cause cancer of the liver, kidneys, pancreas and more.”
http://science.time.com/2011/03/12/japans-radiation-exposure-how-serious-is-it/#ixzz2q8wN3dkO
(2) And Cesium-137 is NOTHING like Potassium in the body.
Humans have evolved to deal potassium in the body.
However, humans have NOT evolved to deal with Cesium-137, which causes cancers, heart disease, etc.
Says the EPA:
“The human body is born with potassium-40 [the type of radiation found in bananas] in its tissues and it is the most common radionuclide in human tissues and in food. We evolved in the presence of potassium-40 and our bodies have well-developed repair mechanisms to respond to its effects. The concentration of potassium-40 in the human body is constant and not affected by concentrations in the environment.”
http://www.washingtonsblog.com/2013/08/are-the-levels-of-fukushima-radiation-hitting-north-america-harmless.html
(3) In the environment, cesium 137 remains for 30 years, which is plenty of time for it to move up the food chain.
(4) Regarding low-level radiation:
“Even the very lowest levels of radiation are harmful to life, scientists have concluded in the Cambridge Philosophical Society’s journal Biological Reviews. Reporting the results of a wide-ranging analysis of 46 peer-reviewed studies published over the past 40 years, researchers from the University of South Carolina and the University of Paris-Sud found that variation in low-level, natural background radiation was found to have small, but highly statistically significant, negative effects on DNA as well as several measures of health.”
http://www.sciencedaily.com/releases/2012/11/121113134224.htm
SilentRunning on Sun, 12th Jan 2014 4:57 am
Keen wrote:
>(1) SilentRunning said: “Cesium, just like Potassium, is bio-excreted and doesn’t build up in the body.”
>Wrong.
Cesium is almost chemically identical to Potassium, and the body handles it in an analogous way. If you look at a periodic table, you will notice that Cs is in column 1, row 6 – directly below Potassium(K) – column 1, row 4.
http://www.webelements.com/
>Here’s the truth:
>Cesium-137 can take years to excrete and meanwhile “that’s more than long enough to cause cancer of the liver, kidneys, pancreas and more.”
Cs-137 can of course cause cancer and kill you above a certain amount. Of that there is no question. I’m not advocating putting it in baby formula. I wish Fukushima had never happened. But the ODDS of it causing you cancer, in the concentrations found in seafood from the Pacific ocean as a whole is very, very small. You stand a far greater risk of being killed in a car accident.
>(2) And Cesium-137 is NOTHING like Potassium in the body.
>Humans have evolved to deal potassium in the body.
Again – that’s wrong – see the periodic table. Also, the “biological half-life” of Cesium is only 70 days.
http://en.wikipedia.org/wiki/Caesium-137
GregT on Sun, 12th Jan 2014 6:30 am
So after spending a bit of time researching the issue of Cesium-137, I found out the following:
The main problem with cesium-137, which is present in the form of salts, is its high water solubility. Cesium-137 ions are readily distributed in the body, and especially in the muscle tissues. The biological half-life is 70 days. This means that half of the cesium-137 has been excreted again after 70 days.
Cesium-137 is cleared by the kidneys which means that it must first be circulating in the blood.
Unlike most other radioisotopes, cesium-137 is not produced from its non-radioactive isotope, but from uranium. It did not occur in nature before nuclear weapons testing began.
There are NO background levels of radioactive cesium or iodine.
The largest single source (of cesium-137 ) was fallout from atmospheric nuclear weapons tests in the 1950s and 1960s, which dispersed and deposited cesium-137 world-wide.
If cesium-137 contaminated soil becomes air-borne as dust, breathing the dust would result in internal exposure. Because the radiation emitting material is then in the body, leaving the site would not end the exposure.
Drinking cesium-137 contaminated water, would also place the cesium-137 inside the body, where it would expose living tissue to gamma and beta radiation.
Compounds accumulate in living things any time they are absorbed and stored faster than they are excreted or metabolized, which means broken down by the body.
Cesium-137 has chemical properties that are similar to potassium. Because the cells in plants, animals and in the human body cannot distinguish between cesium-137 and potassium, cesium-137 can be mistaken by the body to be potassium and absorbed as such. Because most potassium in the human body is found in the blood, cesium-137 can be found in all parts of the human body.